

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء الاشعاعية والنووية
Radical-Chain Addition Reactions to Alkenes
المؤلف:
John D. Roberts and Marjorie C. Caserio
المصدر:
Basic Principles of Organic Chemistry : LibreTexts project
الجزء والصفحة:
........
19-1-2022
7361
Radical-Chain Addition Reactions to Alkenes
The early literature concerning the addition of hydrogen bromide to unsymmetrical alkenes at best is confused. Sometimes the same alkene was reported to give addition both according to, and in opposition to, the principles discussed for electrophilic ionic addition . Much of the uncertainty on the addition of hydrogen bromide was removed by the classical researches of M. S. Kharasch and F. R. Mayo (1933) who showed that there must be two reaction mechanisms, each giving a different product. Kharasch and Mayo found, in the presence of radical inhibitors, hydrogen bromide adds to propene in a rather slow reaction to give pure 2-bromopropane:

With light, peroxides, radical initiators, and in the absence of radical inhibitors a rapid radical-chain addition of hydrogen bromide occurs to yield 80% or more of 1-bromopropane:
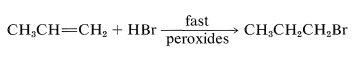
Similar effects have been noted occasionally with hydrogen chloride, but never with hydrogen iodide or hydrogen fluoride. A few substances apparently add to alkenes only by radical mechanisms, and always add in the opposite way to that expected for electrophilic ionic addition.
Two questions with regard to the so-called abnormal addition will be given special attention. Why does the radical mechanism give a product of different structure than the ionic addition? Why does the radical addition occur readily with hydrogen bromide but rarely with the other hydrogen halides?
The abnormal addition of hydrogen bromide is catalyzed strongly by peroxides, which have the structure R−O−O−R and decompose thermally to give RO⋅ radicals:

The RO⋅ radicals can react with hydrogen bromide in two ways, to abstract either hydrogen atoms or bromine atoms:
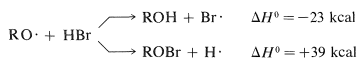
Clearly, the formation of ROH and a bromine atom is energetically more favorable. The overall process of decomposition of peroxide and attack on hydrogen bromide, which results in the formation of a bromine atom, can initiate a radical-chain addition of hydrogen bromide to an alkene.
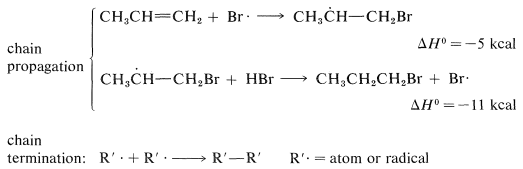
The two chain-propagating steps, taken together, are exothermic by 16kcal and have a fairly reasonable energy balance between the separate steps. The reaction chains apparently are rather long, because the addition is strongly inhibited by radical traps and only traces of peroxide catalyst are needed.
 الاكثر قراءة في المركبات الوسطية وميكانيكيات التفاعلات العضوية
الاكثر قراءة في المركبات الوسطية وميكانيكيات التفاعلات العضوية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















